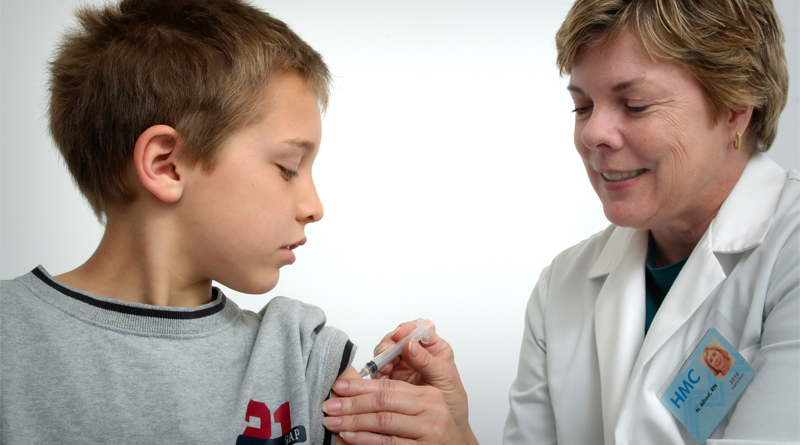
Children ages 2 to 11 could access the COVID-19 vaccine this fall
DORAL, FL – Children ages 2 to 11 could access Pfizer’s COVID-19 vaccine in September, said the pharmaceutical company’s chairman and chief executive officer, Dr. Albert Bourla.
During a quarterly earnings call, Bourla announced the company hopes to request emergency use authorization from the U.S. Food and Drug Administration this fall aimed at providing the vaccine to the youngest population.
In addition, following the approval of the vaccine for children ages 2 to 11, Pfizer could also request an emergency use authorization for ages six months to 2 years in the fourth quarter of the year.
But children ages 12 to 15 could become the first in line much sooner than that. The Food and Drug Administration is expected to authorize Pfizer’s vaccine later this week or early next week to this age range.
Both Pfizer and its partner BioNTech reported in March their vaccine is safe and 100% effective in this age group. According to their preliminary results from a study of 2,260 U.S. volunteers ages 12 to 15, there were no cases of COVID-19 among fully vaccinated children compared with 18 among those given dummy shots.
President Joe Biden said the administration is prepared to ship doses to 20,000 pharmacies across the nation once the approval for this age group is granted.
“I want American parents to know that if that announcement comes, we are ready to move immediately,” Biden said.
Vaccines will become available through pediatricians and family physicians and the goal is for states to vaccinate as many teens as possible by July 4. This day is also the deadline the government has set to immunize against COVID-19 at least 70% of the adult population with one dose.
“Parents and their children can talk to their family doctor about it and get their shot from a provider they trust the most. Easy, fast and free,” Biden said. “And if teens are on the move this summer, they can get their first shot in one place and the second shot elsewhere.”
The efforts to make children eligible for the COVID-19 vaccine are to reduce the number of infections across the nation and among the younger populations. Just last week, children accounted 22.4% of new cases.
In addition, pediatric vaccines are intended to make the upcoming school year go smoother than the current one disrupted by online learning, lockdowns of certain classrooms and changes in events’s logistics.
Photo: Unsplash.com